Longer interval between maternal RSV vaccination and birth increases placental transfer efficiency
TAP was employed to collect capillary blood from 2-month-old infants for antibody tests of RSV and pertussis toxin.
Read More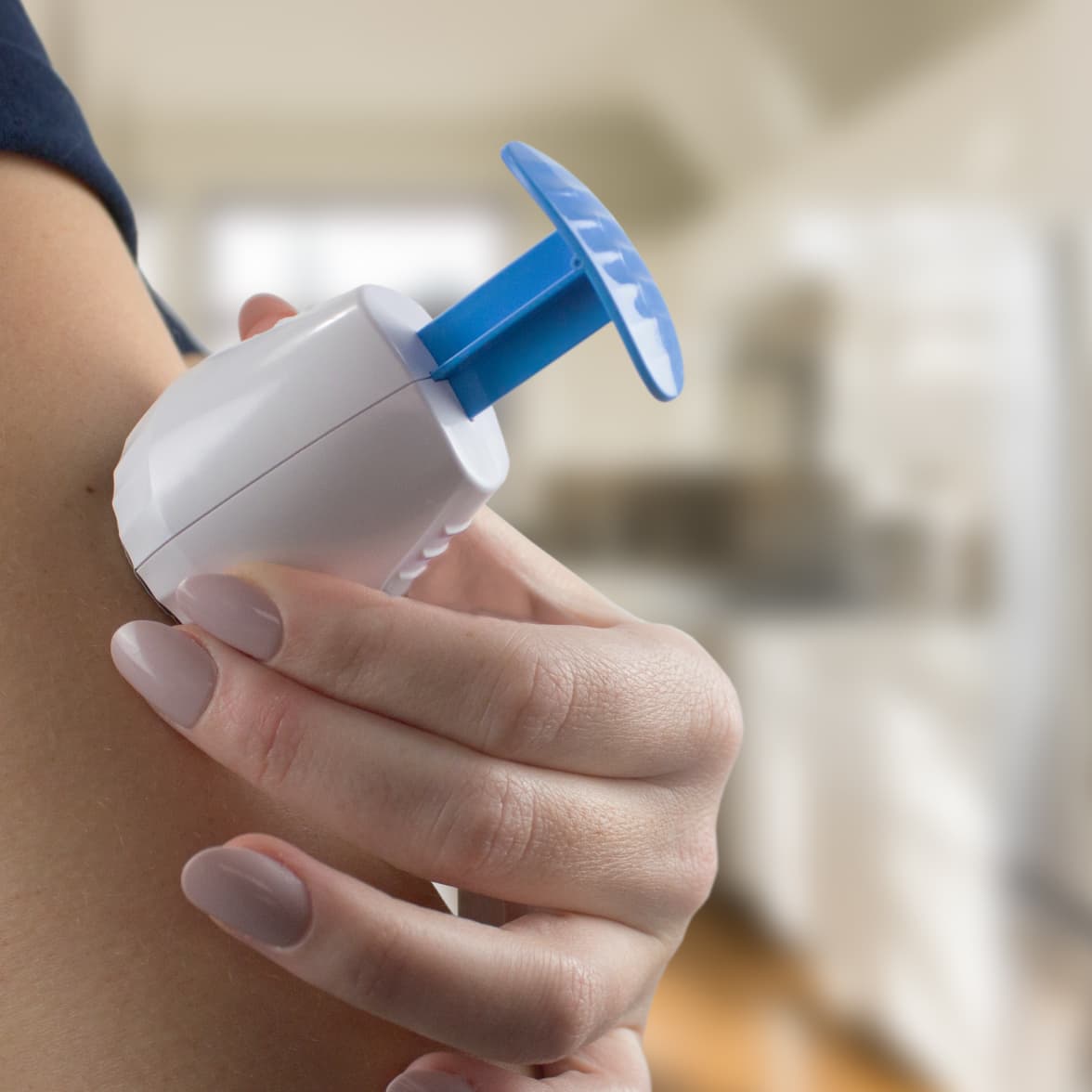
Introducing TAP® Micro and TAP® Micro Select
Our Touch Activated Phlebotomy (TAP®) devices have been developed over the last 15 years to enable virtually painless, remote blood collection.
Our devices produce high-quality, whole blood samples at consistently high volumes of up to 900µL through virtually painless capillary blood collection, making them ideal for decentralized clinical trials, wellness testing, and clinical-grade blood sampling. Remote blood collection allows the opportunity to replace traditional phlebotomist-conducted venous blood collection or painful fingerstick tests in at home blood collection applications.
TAP® Micro Select uses the same bladeless blood draw technology as TAP® Micro and takes the pain out of blood collection in decentralized clinical trials as well as health and wellness testing. Using HALO™ technology, a bladeless microneedle array, TAP® Micro Select enables virtually painless, large-volume collection of up to 900µL of whole, capillary high-quality blood.
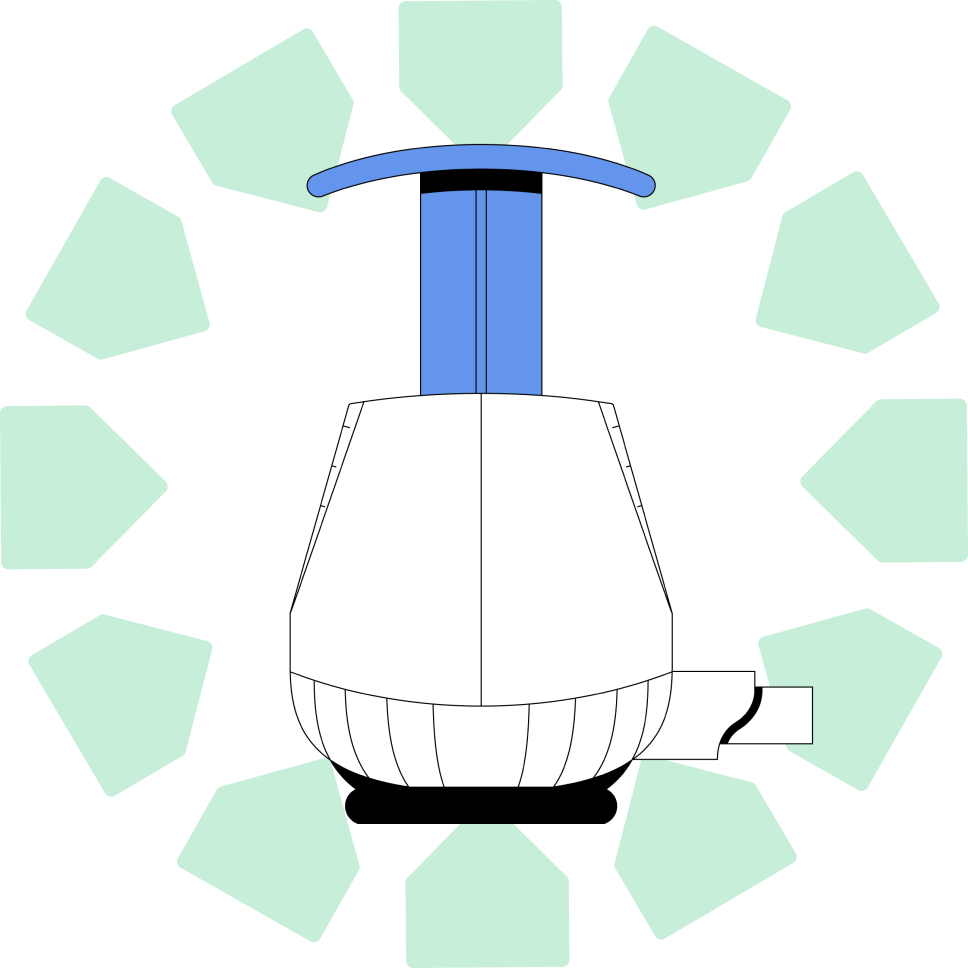
TAP® Micro offers an easy-to-remove detachable sample tube, with capillary blood collection volumes of as much as 900µL, and multiple sample tube type choices to meet customer needs. TAP® Micro devices have been successfully employed by hospitals and medical professionals for investigational use to conduct remote blood collection on a wide range of patients spanning ages and conditions, validating its flexibility and effectiveness in the field.
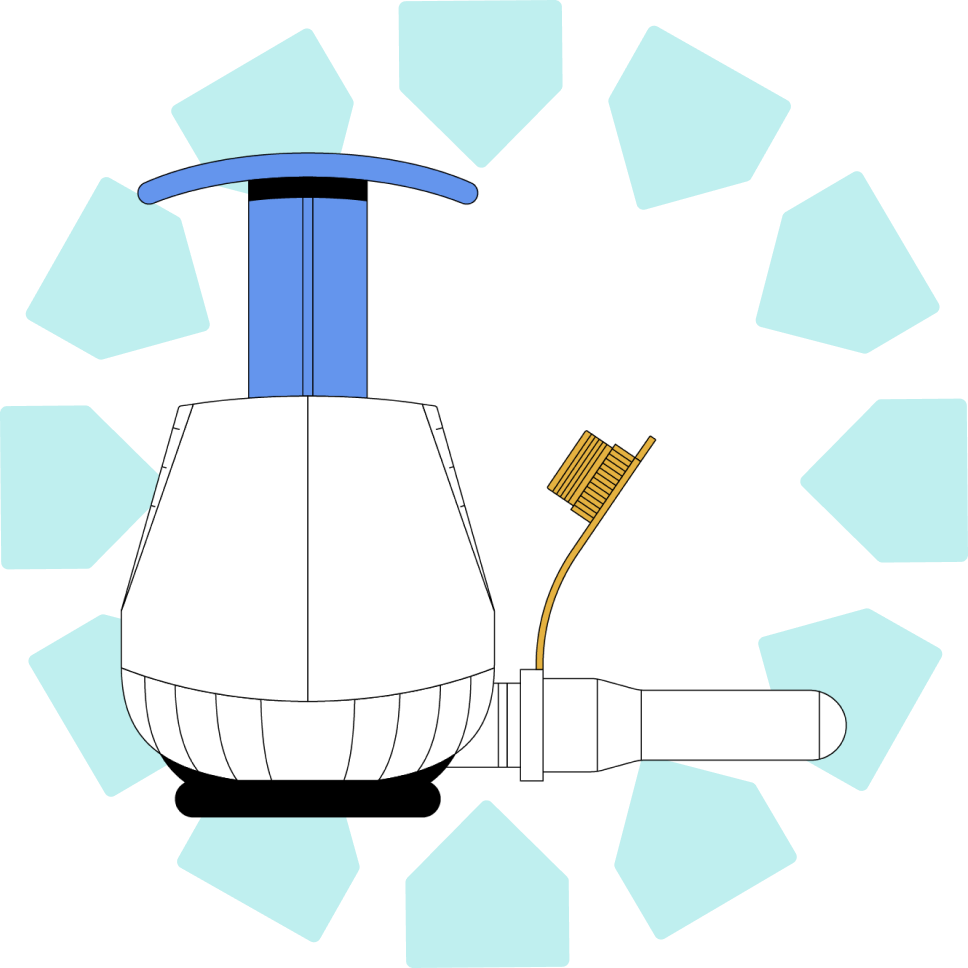
We offer customizable sample management services, including kitting, fulfillment, and sample tracking from the end user to any central lab.