-
Whitepaper
TAP® technology shows promise for early, precise Alzheimer’s monitoring via biomarkers
Quanterix Simoa HD-X Immunoassays
HIGHLIGHTS
- The TAP is a minimally invasive and virtually painless blood collection device that enables point-of-care, capillary whole blood collections for downstream proteomics applications such as multiplexed immunoassays.
- The Quanterix Simoa HD-X platform enables an ultrasensitive, fully automated immunoassay platform with multiplexing detection at fg/ml concentrations, dynamic ranges at >4 logs, and precision at <10% CVs using bead-based single-molecular arrays (Simoa).
- The Biomarker Assay Laboratory (BAL) at the National Centralized Repository for Alzheimer’s Disease and Related Dementias (NCRAD) incorporates a standardized process for Quanterix Simoa assessments for AD biomarkers including Abeta 40 (AB-40), Abeta 42 (AB-42), Glial Fibrillary Acidic Protein (GFAP), Neurofilament light (NfL) chain, and an isoform of phosphorylated tau protein (P-tau 181).
- Analytical concordance was assessed for Quanterix Simoa HD-X outputs for donor samples between TAP vs traditional venipuncture collections.
- Applications for efficient blood collection coupled to ultrasensitive proteomics can enable early disease indication and monitoring utility.
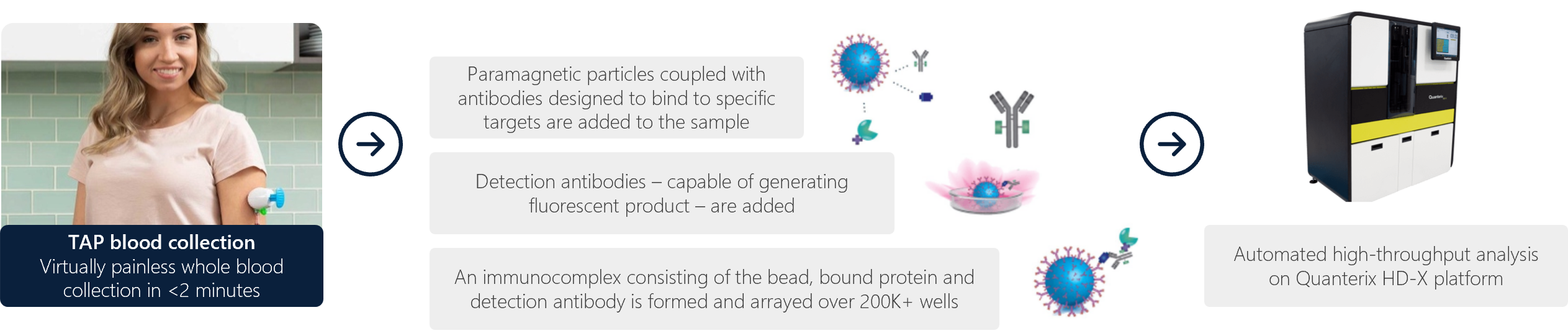
Figure 1: Overall workflow showing TAP blood collection and high-level Quanterix Simoa workflow.
INTRODUCTION
Reliable and ultrasensitive detection of biomarkers is desired for early detection, prevention and monitoring strategies for understanding disease pathology and intervention. Plasma biomarkers are particularly attractive as a minimally invasive prognostic indicator for early intervention and highlights the need for ultrasensitive assays in AD patients. For comparison, PET brain imaging and CSF sampling are generally conducted as symptom-based tests, which are invasive, expensive and limited in access. Therefore, routine blood tests for plasma markers in AD enable cost-effective paradigms for patient screening and monitoring.
The Quanterix Simoa HD-X enables on average 1000-fold increase in sensitivity as compared to traditional immunoassays like ELISAs. The bead-based array platform incorporates Simoa (single molecular array) technology to assess individual analytes at scale. Technologies that enable specific, ultrasensitive and targeted biomarker interrogations from microsample volumes with low costs per datapoint enable routine monitoring of timepoints for disease stratification between AD and non-AD individuals.
Traditional blood collection for plasma assessments involve a venipuncture draw by a trained phlebotomist, whereby 5-10 ml of whole blood is collected in a standard citrate/heparin/EDTA tube.
This procedure can be relatively costly and inaccessible to scale across large patient populations for screening purposes, whereby economic and logistical hurdles to blood collections may persist due to the need of a trained phlebotomist. Here, we introduce the Yourbio Health TAP blood collection device as a viable tool to enable feasibility of blood collection scale and adoptability for downstream Quanterix Simoa HD-X proteomics interrogations. The TAP enables point-of-care collections by novice users, direct use by physicians/technologists in general practices, or collections in underserved/remote geographies. We obtained paired blood samples (TAP + traditional venipuncture) to analyze downstream Quanterix Simoa HD-X biomarkers implicated in AD to analytical feasibility.
SELECTION OF ALZHEIMER’S DISEASE PLASMA BIOMARKERS FOR TAP EVALUATIONS
We selected 5 plasma biomarkers per NCRAD recommendation related to AD: AB40, AB42, GFAP, NF-L, P-tau 181. In general, elevated plasma concentrations of these biomarkers are implicated in plaque accumulation and deposition, abnormal phosphorylation and filament bundle aggregation, and neuronal degeneration for overall AD progression.
- Amyloid beta peptides – Amyloid-B40 (AB40) and Amyloid-B42 (AB42): Amyloid beta peptide accumulation in the brain can form extracellular plaques. AB40 and AB42 are the two primary forms of amyloid beta that are generated from amyloid precursor protein. AB40 is the most abundant of the two forms in both health and disease states (~10-20x higher concentrations than AB42). Therefore, the AB42:AB40 ratio is a useful metric to stratify between AD and non-AD individuals in addition to AD progression as a routine monitoring timepoint. The low concentrations of AB peptides in plasma (~100x lower in plasma than in the brain/CSF) make the Quanterix Simoa a practical choice for ultrasensitive detection.
- Glial fibrillary acidic protein (GFAP): GFAP is a filament protein that can polymerize with other proteins as well as forming homo- and hetero-dimers in the astrocytic glial cell environment within the central nervous system. High plasma GFAP concentrations have been shown to predict the clinical progression of AD pathology.
- Neurofilament Light (NF-L): Expressed only in neurons as a cytoskeletal filament (along with neurofilament medium and neurofilament heavy), elevated NF-L is associated with neurodegeneration and AD progression as a result of axonal damage.
- Plasma tau 181 (P-tau 181): P-tau 181 is one of six major isoforms that is elevated in neurodegenerative diseases. Abnormal phosphorylation of tau (in this case, at residue 181) enables aggregation of filament bundles. While most tau is localized to the CNS, peripheral tau is of low abundance in plasma. Elevated p-tau 181 has been associated with 3.5x increase in AD individuals, cognitive decline and gray matter loss, making it a viable biomarker for ultrasensitive detection methods. (Note that p-tau 217 and p-tau 231 are emerging as more sensitive biomarkers within plasma for AD stratification.)
- Amyloid, Tau, Neurodegeneration (ATN) Classification: The ATN framework enables AD risk stratification based on the selected biomarkers for classifying amyloid pathology (A), tau pathology (T) and neurodegeneration (N). These binary categories are scored as positive or negative (example: A+, T+, N- or A-, T+ N+, etc). The table to the right stratifies risk of short-term cognitive decline based on the biomarker profile and cognitive stage.
Figure 2: ATN classification system from NIA-AA Research
Framework (Jack et al. 2018.)
MATERIALS AND METHODS
Under an IRB-approved protocol, 18 donors (seemingly normal; without AD) had paired blood samples collected by a trained phlebotomist (traditional venipuncture tube) and assisted-collections (TAP device) into heparin-coated tubes. The venipuncture tubes collected the full max fill volume of 3 ml by the phlebotomist, whereas the TAP devices collected at least 300 ul and up to 600 ul. All blood samples were collected and treated identically (next-day shipment to lab on dry ice during transit) in preparation for downstream proteomics. Briefly, all whole blood collections were centrifuged to obtain plasma, whereby 200 ul of the resulting plasma (each for venipuncture and TAP collections) was used as input volume for the Quanterix Simoa HD-X assessments (100 ul for AB42/AB40/NF-L/GFAP and 100 ul for p-tau 181). Wet-lab setup was performed according to NCRAD’s and Quanterix Simoa HD-X SOPs.
Data analysis was conducted whereby each donor sample method (VP + TAP) was run on two Quanterix Simoa assays: N4PE multiplex (AB42/AB40/NF-L/GFAP) and p-tau 181 singleplex. Each sample per assay was run in duplicate to report 36 mean concentration values, whereby the mean, standard deviation, and %CV were assessed. Concentrations, log transformations of concentrations, best-fit values, and Bland-Altman plots were generated to understand bias between both sample collection methods for analytical assay feasibility.
RESULTS
A summary data table for all donors (n=18) shows individual results calculated from duplicates per sample for all 5 AD biomarkers. Data for intra-assay %CV, Pearson correlation, average bias with 95% CI, percent difference within donors, descriptive statistics, regression statistics and a t-test (paired two sample) is presented to show statistical comparability between TAP vs VP samples. Correlation is the intended primary outcome for this initial feasibility study.
Overall, results show generally positive correlation between TAP and VP samples for all tested analytes except P-tau 181. The overall intra-assay precision for P-tau 181 is <10% while bias is ~185%. Possible explanations may be related to biological differences of the target within venous vs. capillary environments and that seven isoforms of P-tau exist (181 was chosen for this study, whereas 217 and 231 have recently emerged as more sensitive isoforms for testing). While correlation for the individual amyloid beta proteins are modest, the ratio demonstrates higher correlation for this metric (which is the intended measure). This may be suggestive of donor-to-donor variability of independent AB proteins, whereby such algorithmic scoring for complex proteins may be necessary for accurate measurements. Correlation for GFAP is very strong (0.9681) as compared to the other bioamarkers, which may indicate that protein-specific correlations should be considered per biomarker between the collection types.
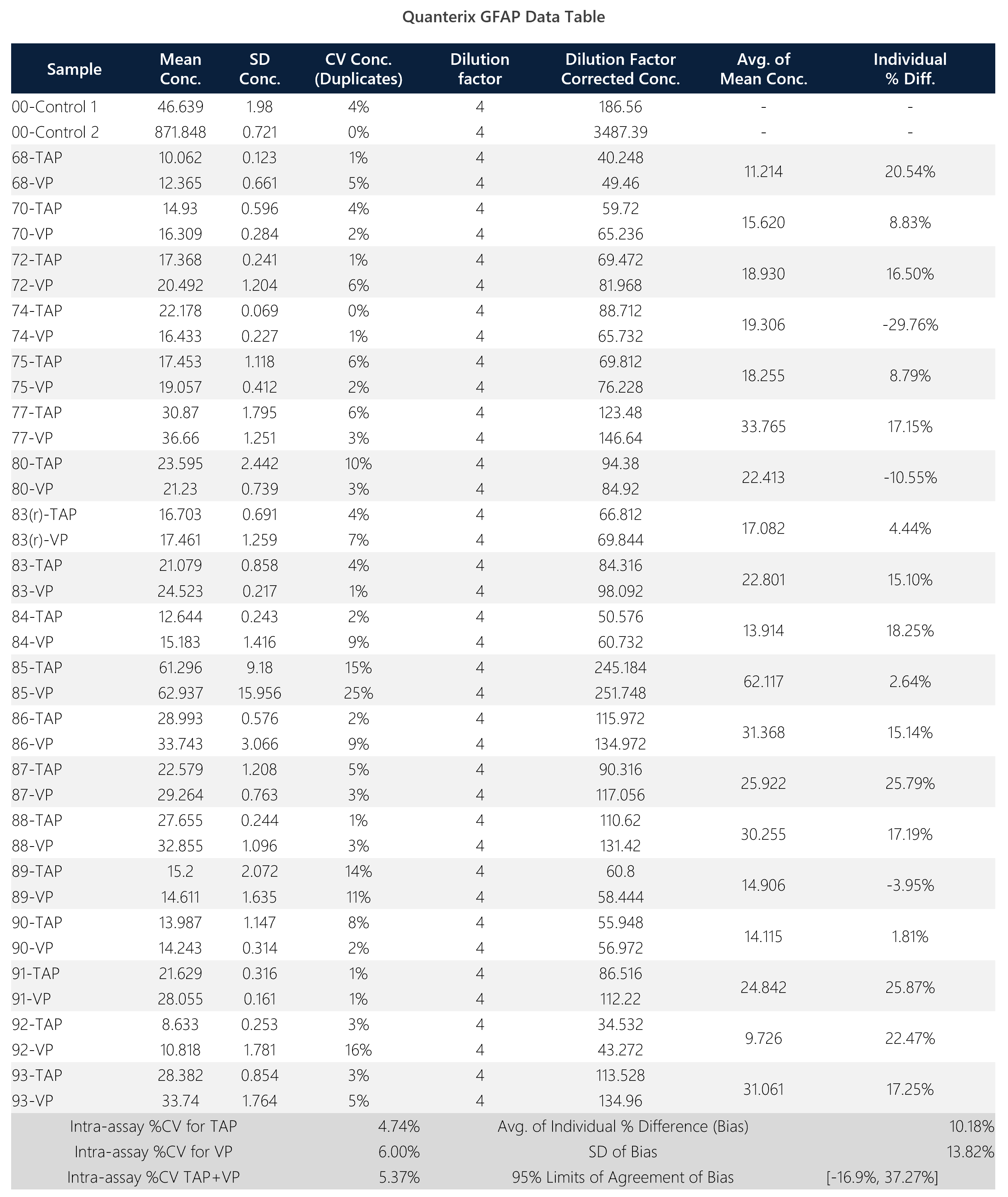
Table 1: Summary data table for Quanterix GFAP assay.
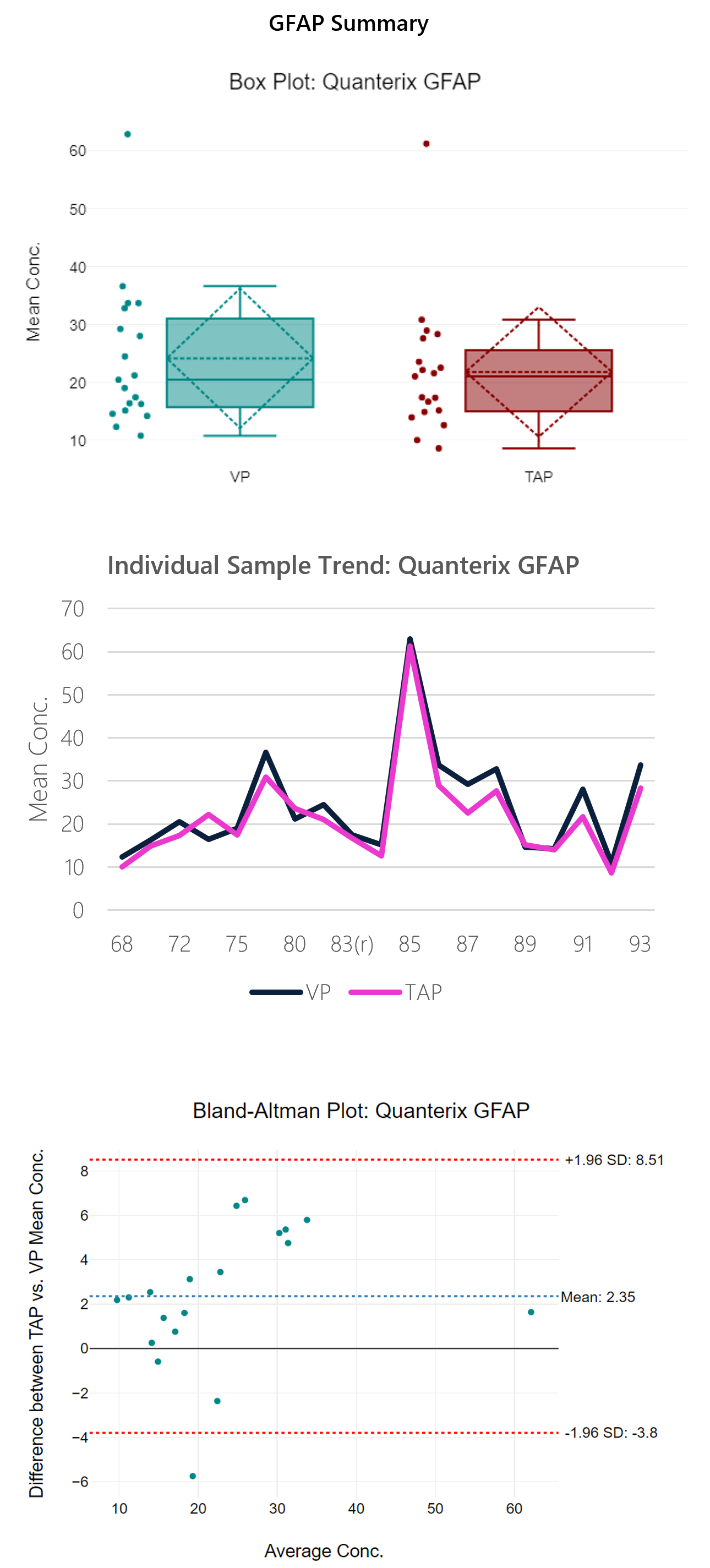

Figure 3: Box plot, sample trend, best fit model and Bland-Altman plots with descriptive, t-test and regression statistics. Overall, the run shows good correlation (0.97) with a y-intercept of 0.06.
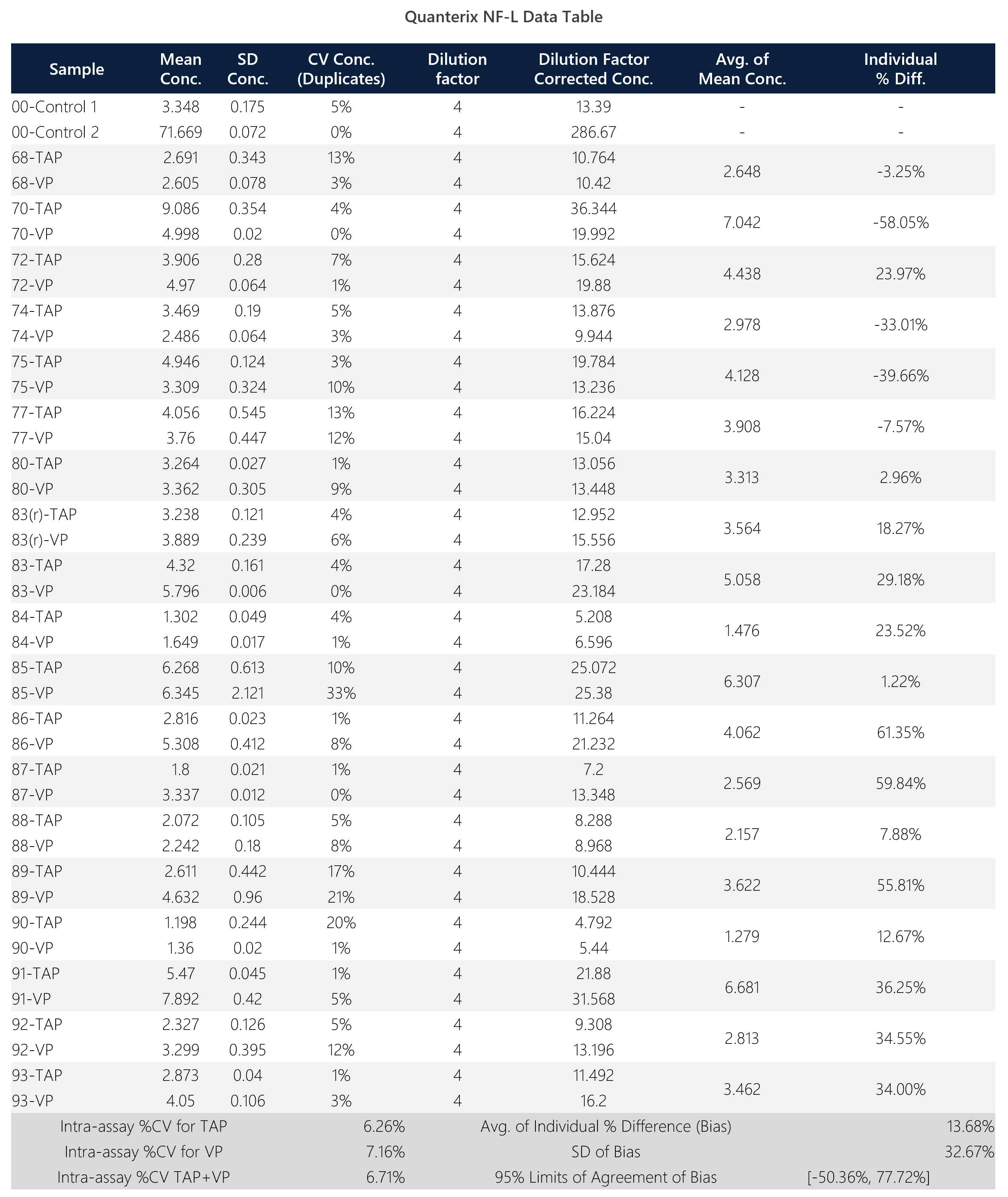
Table 2: Summary data table for Quanterix NF-L assay.
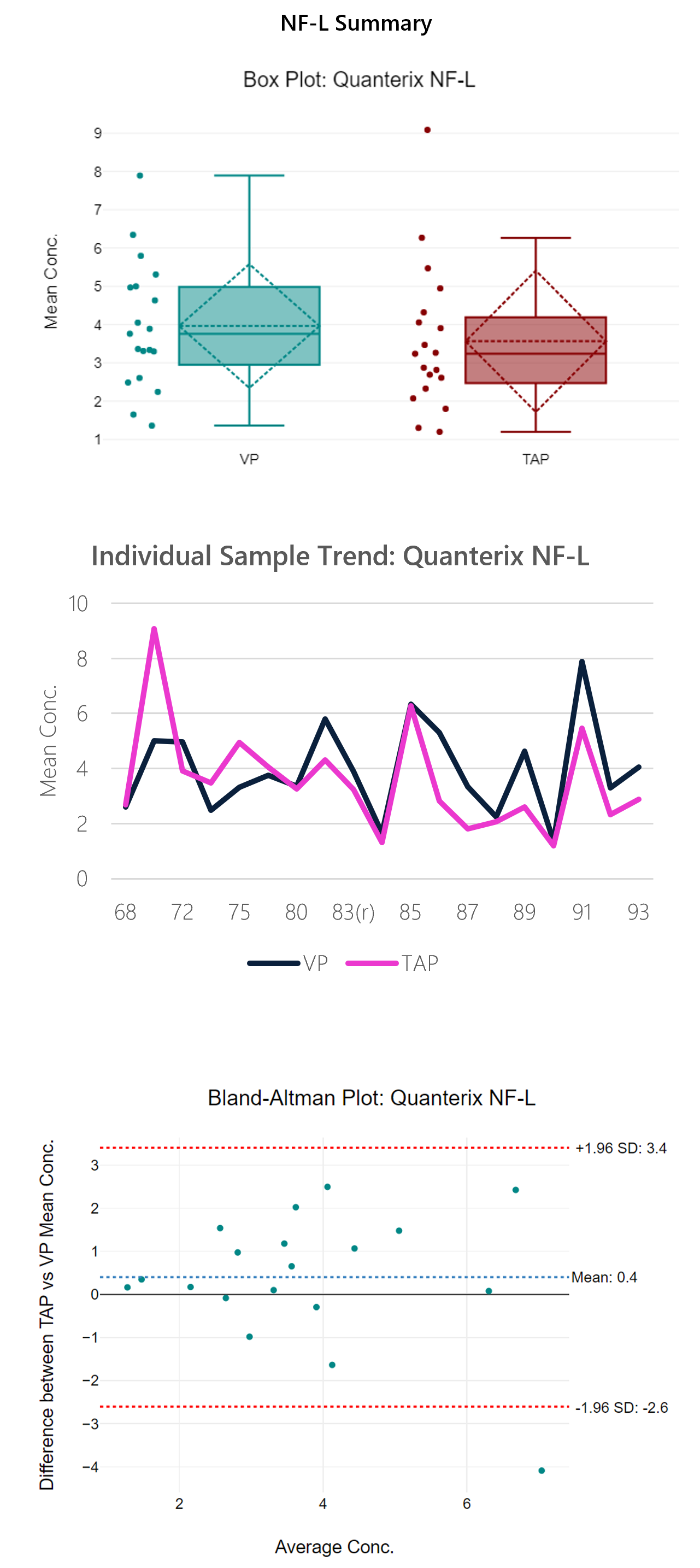

Figure 4: Box plot, sample trend, best fit model and Bland-Altman plots with descriptive, t-test and regression statistics. Overall, the run shows modest correlation (0.64) with a y-intercept of 0.68.
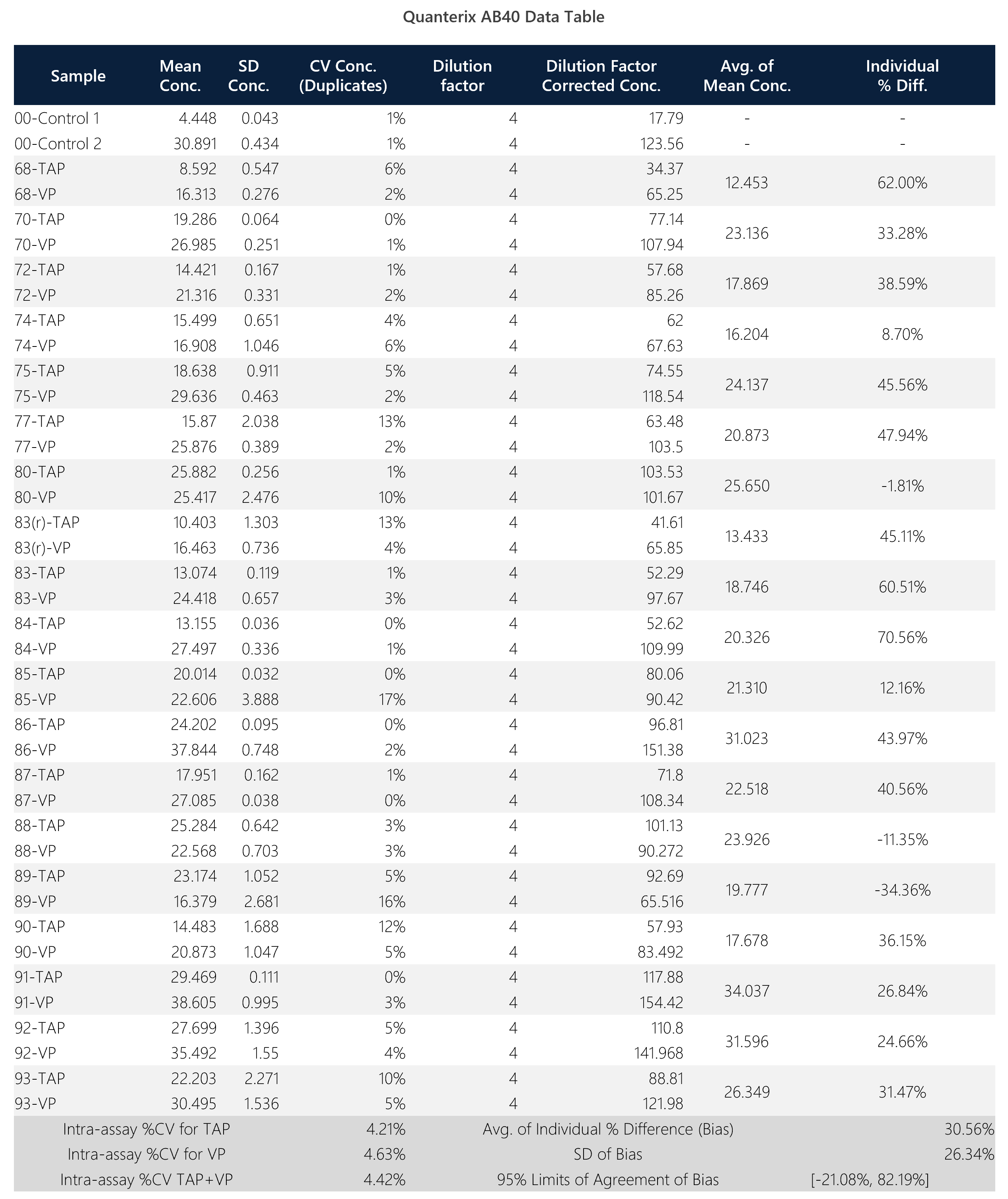
Table 3: Summary data table for Quanterix AB-40 assay.
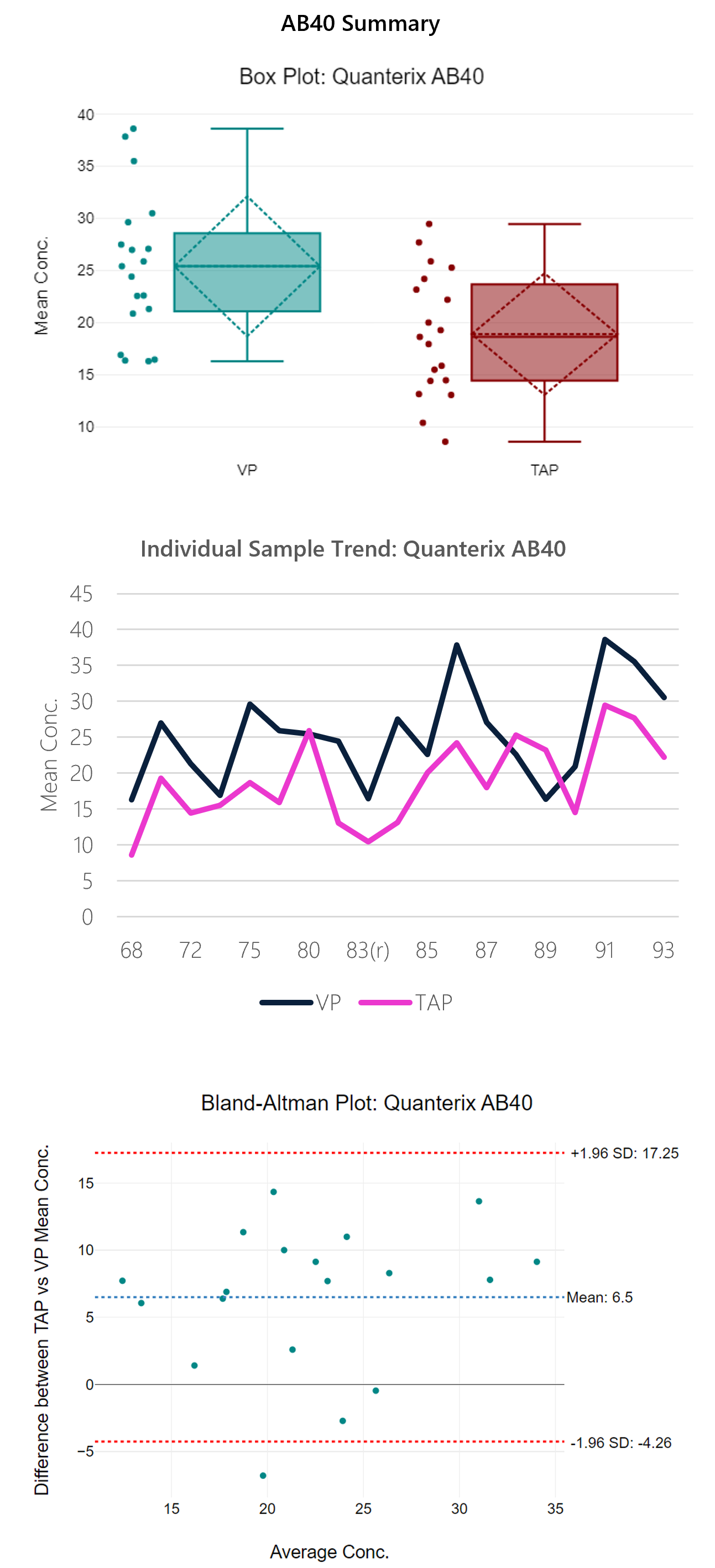

Figure 5: Box plot, sample trend, best fit model and Bland-Altman plots with descriptive, t-test and regression statistics. Overall, the run shows modest correlation (0.65) with a y-intercept of 4.69.
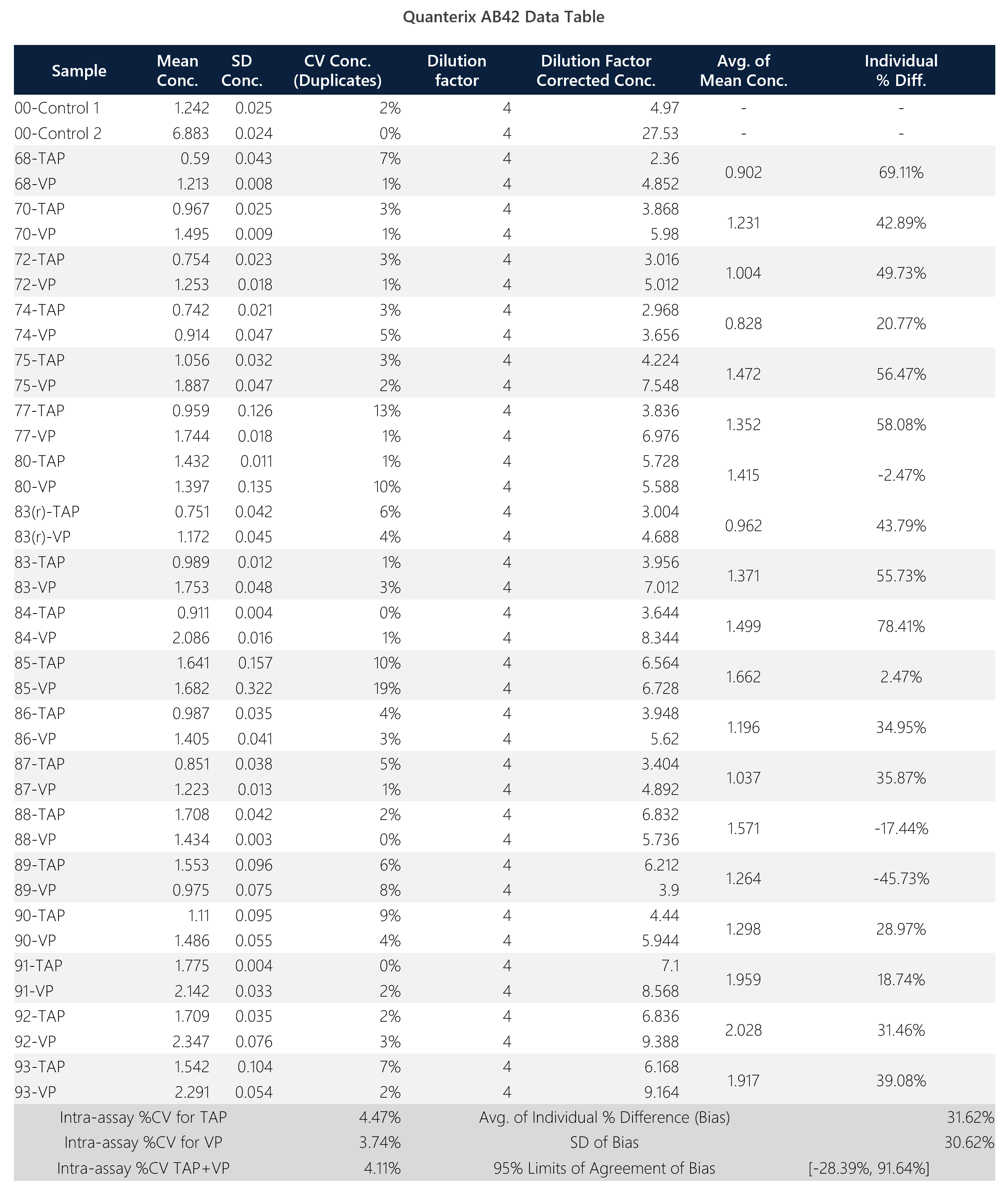
Table 4: Summary data table for Quanterix AB-42 assay.
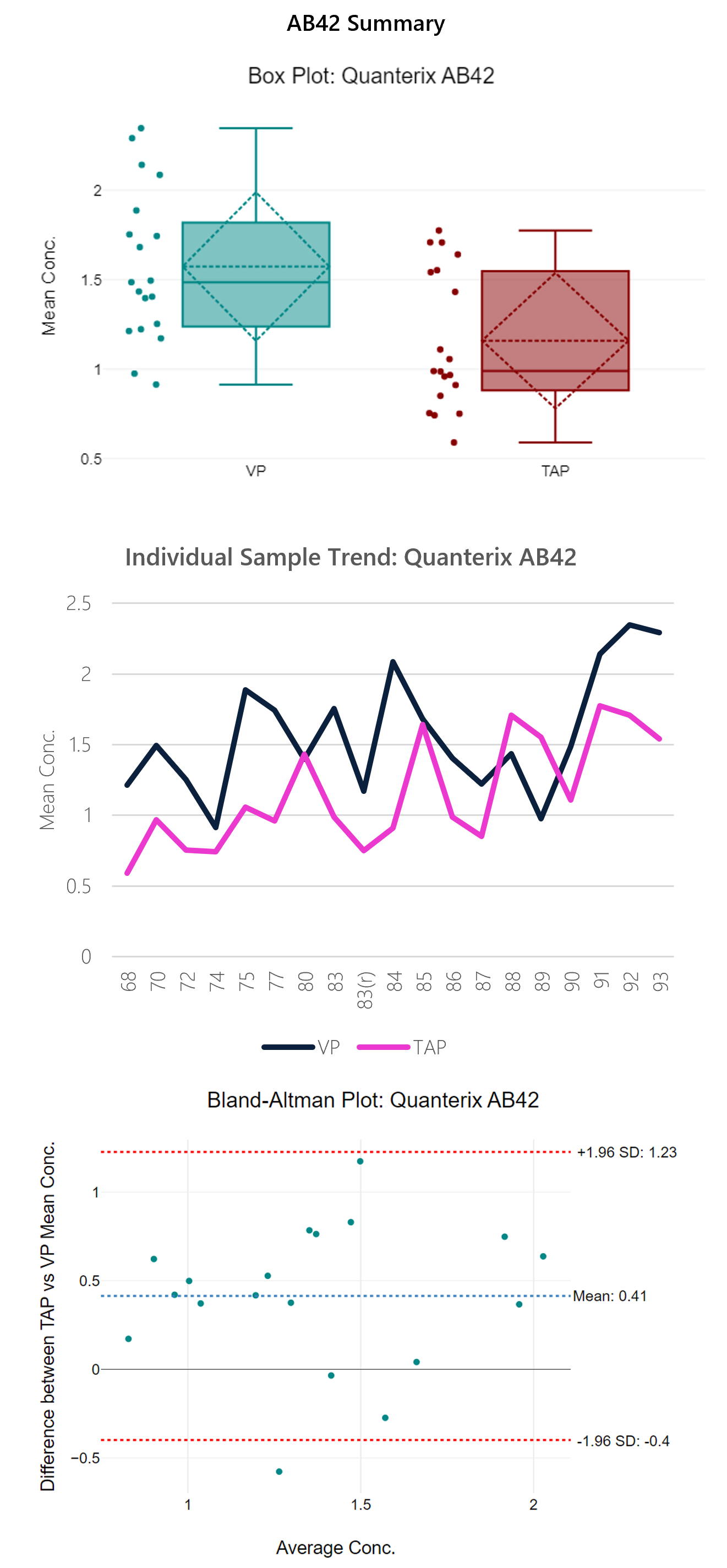

Figure 6: Box plot, sample trend, best fit model and Bland-Altman plots with descriptive, t-test and regression statistics. Overall, the run shows modest correlation (0.49) with a y-intercept of 0.46.
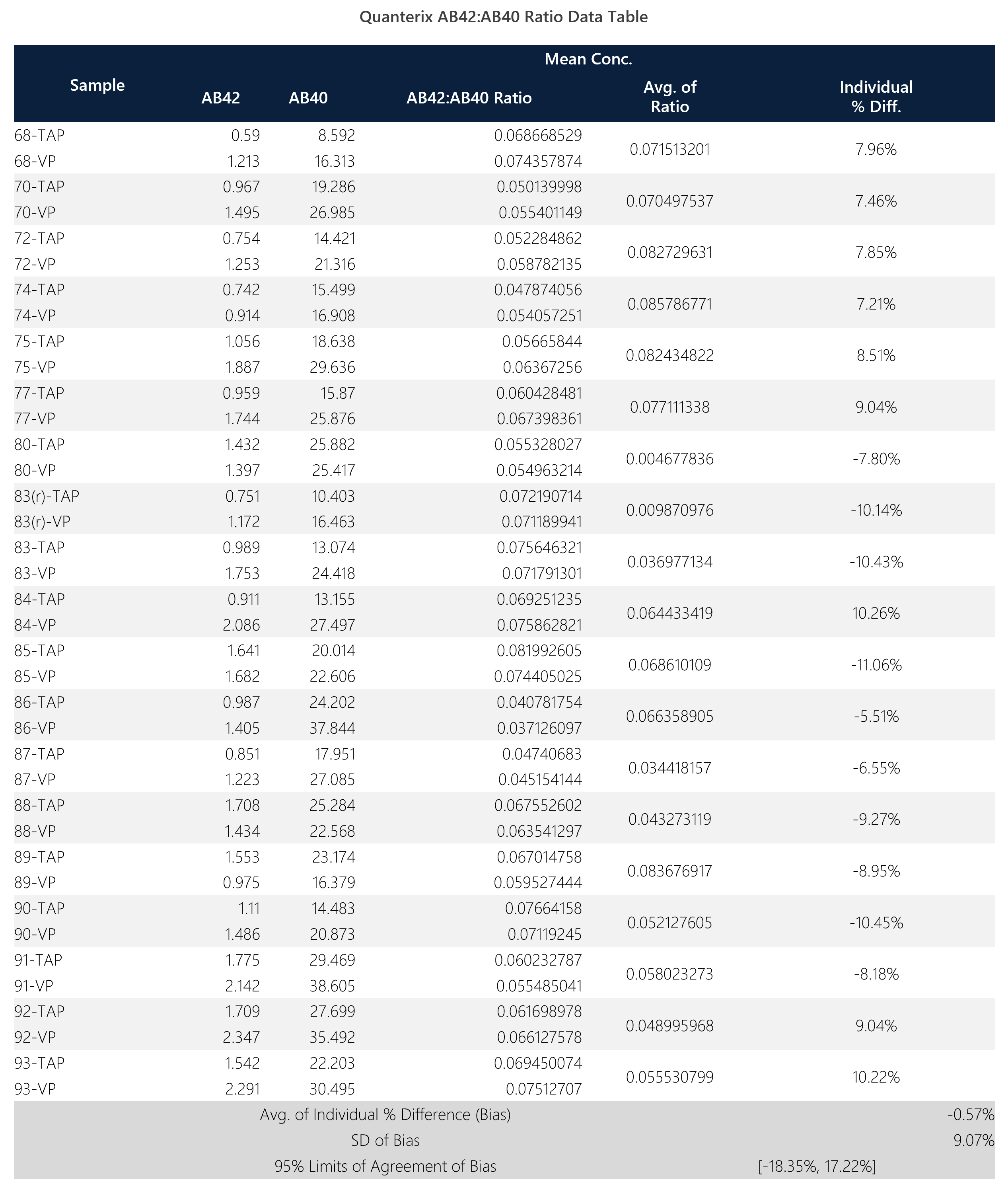
Table 5: Summary data table for Quanterix AB-42:AB-40 ratio.
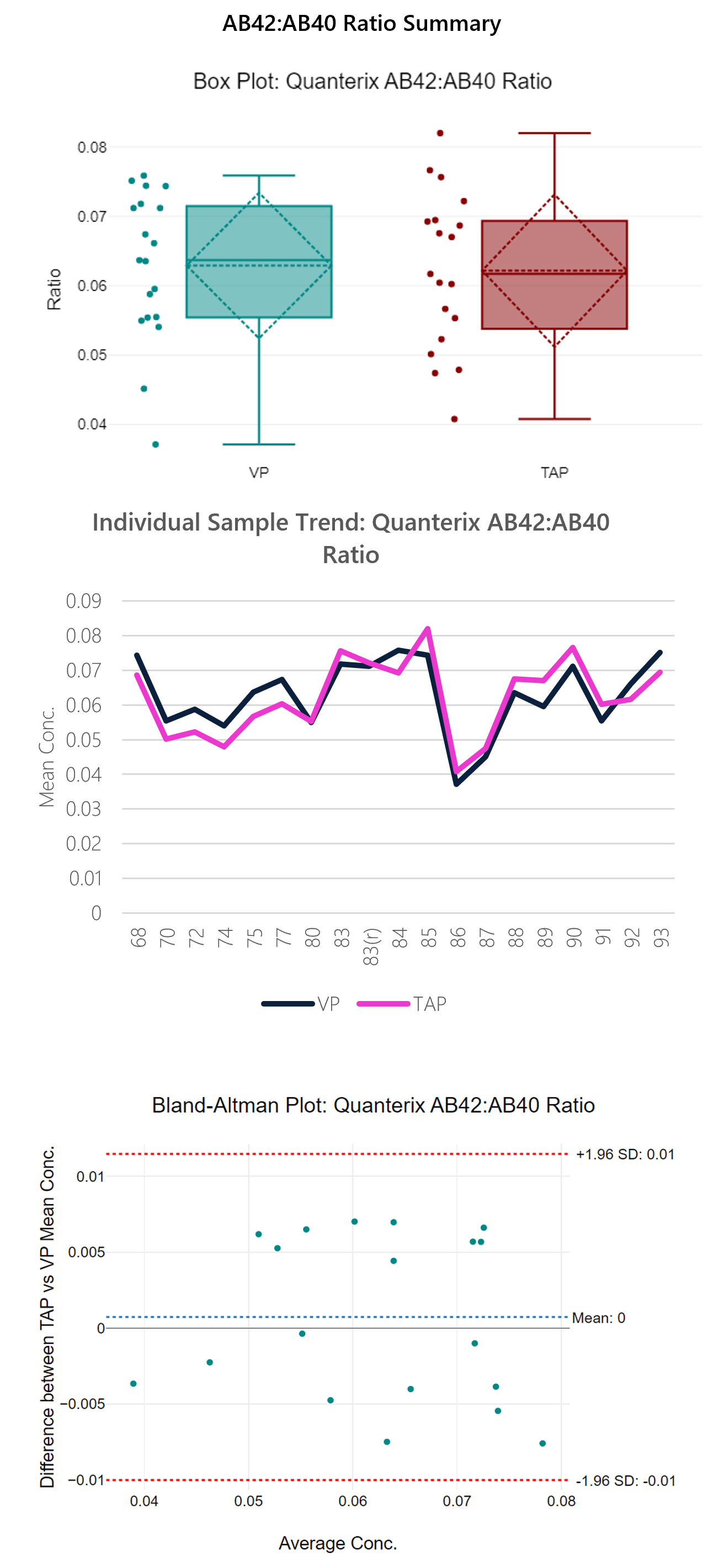
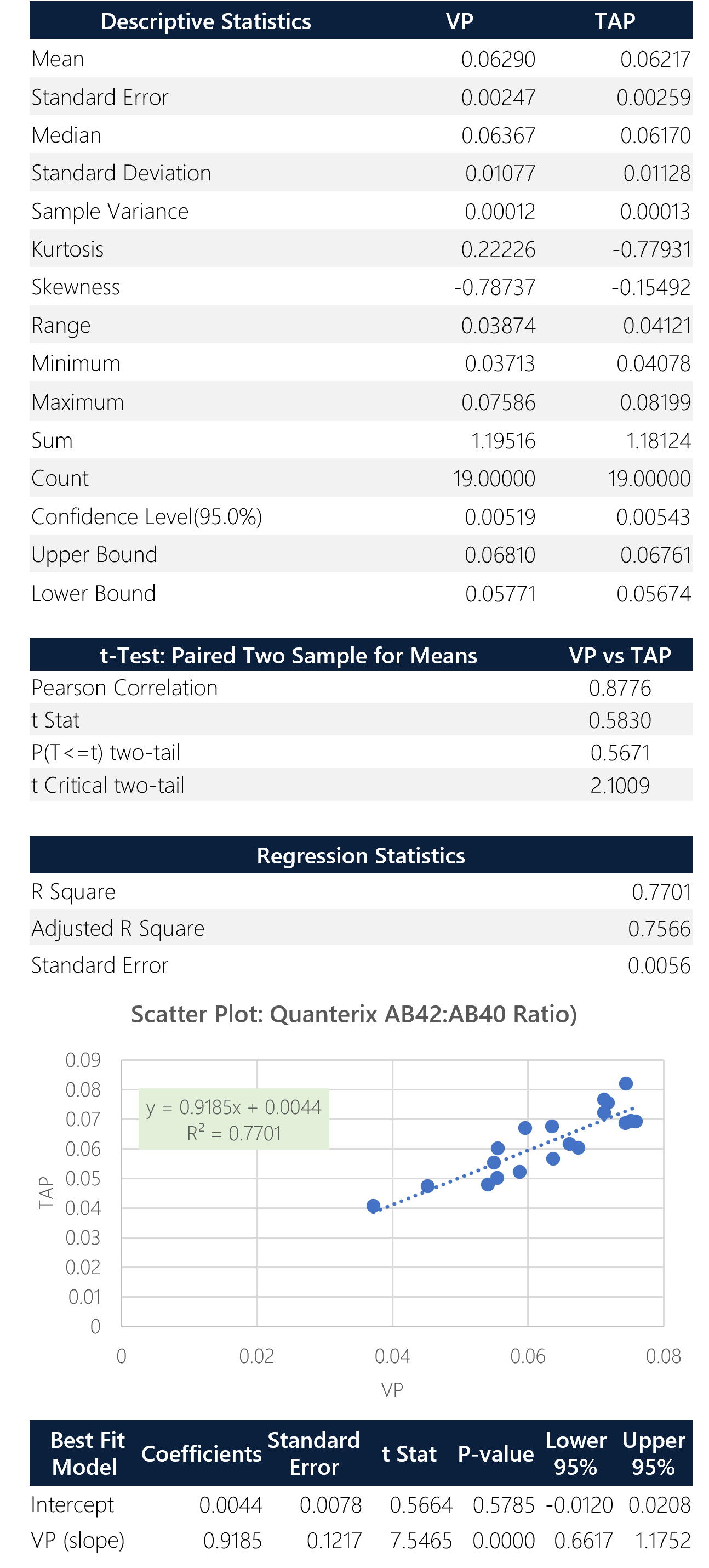
Figure 7: Box plot, sample trend, best fit model and Bland-Altman plots with descriptive, t-test and regression statistics. Overall, the ratio shows good correlation (0.88) with a y-intercept of 0.0044.
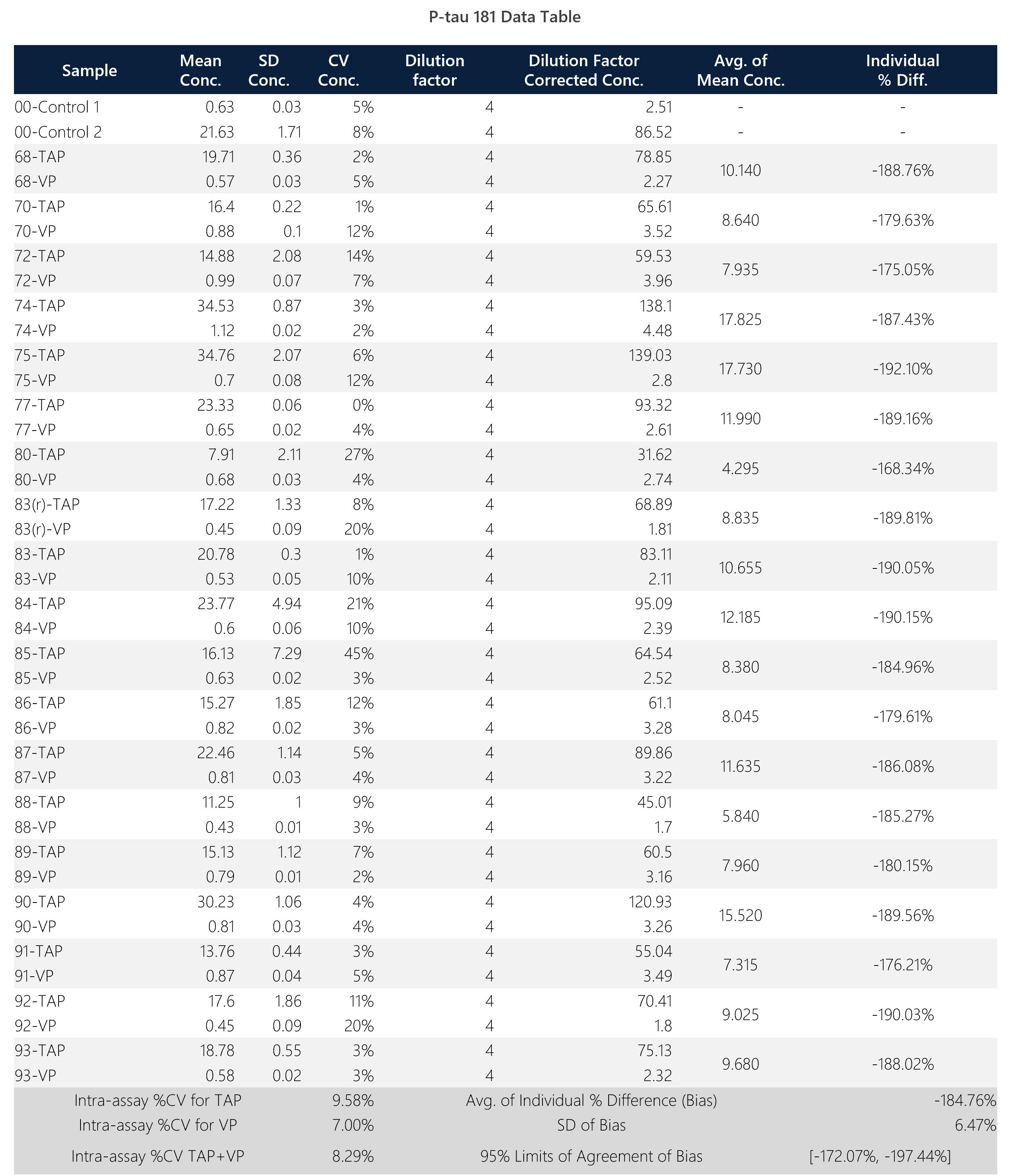
Table 6: Summary data table for Quanterix p-tau 181 assay.
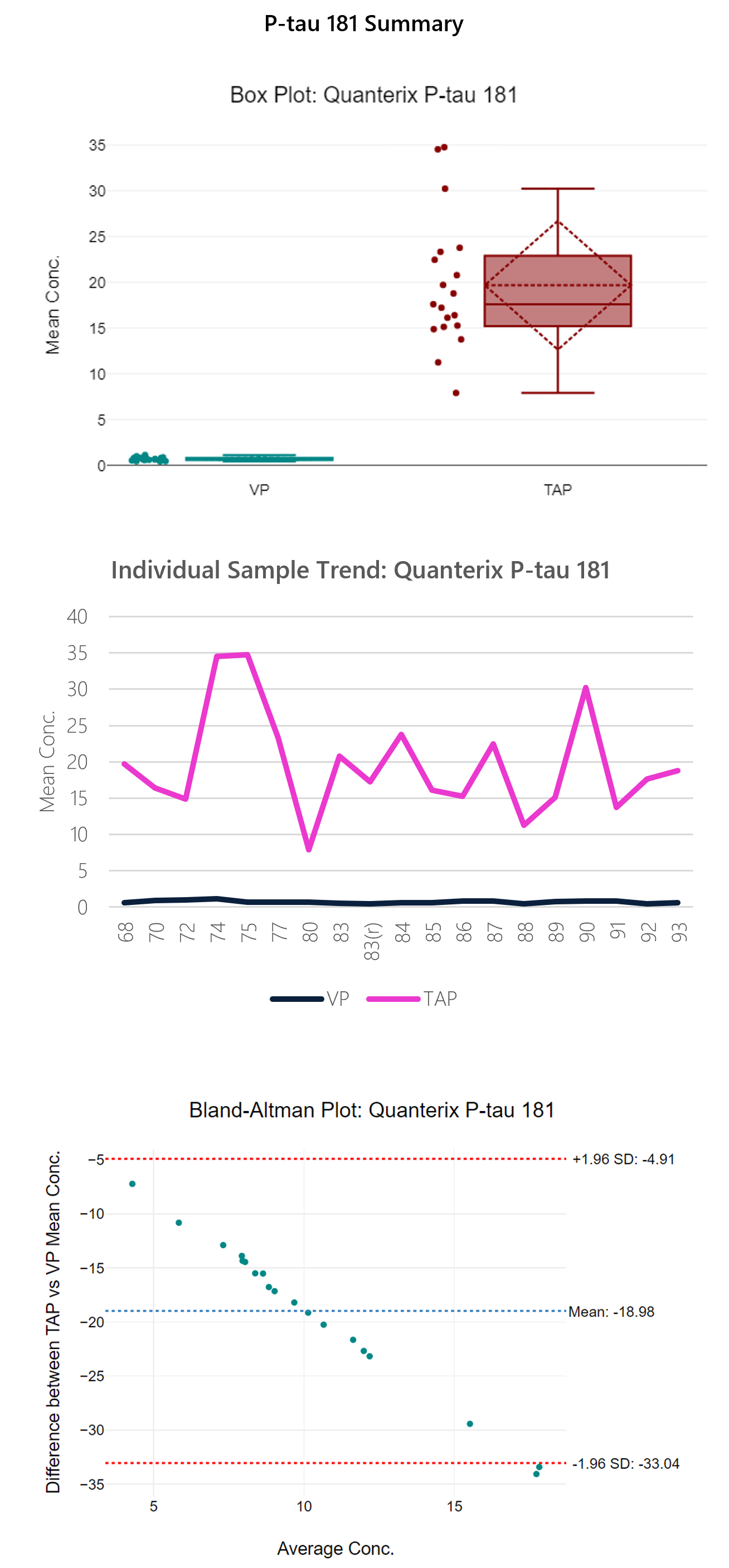
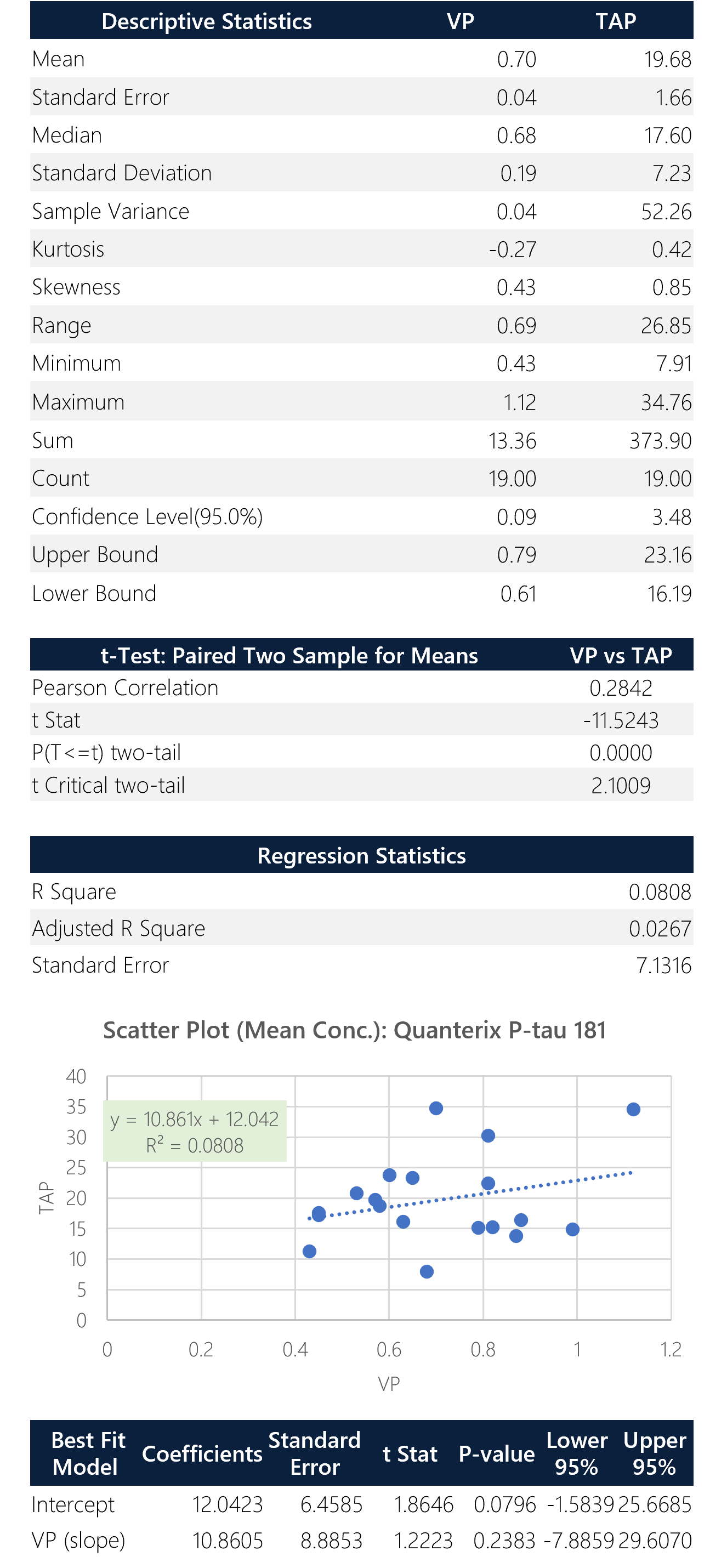
Figure 8: Box plot, sample trend, best fit model and Bland-Altman plots with descriptive, t-test and regression statistics. Overall, the p-tau 181 isoform shows poor correlation (0.28) with a y-intercept of 12.04. This assay shows significant bias (185%) between TAP and VP.
CONCLUSIONS
Accessible and minimally invasive sampling with the TAP device enables testing of AD biomarkers over more frequent timepoints. While most of the intended markers show generally good correlation, variable differences between biomarkers exist within the AD-related assays. Specifically, p-tau 181 isoform shows significant bias and other isoforms should be considered. Remote monitoring can enable better data analysis when coupled to sensitive and specific platform technologies such as Quanterix assays. The purpose of this study is to demonstrate technology and platform capability of the TAP device with the Quanterix Simoa HD-X assays. Further evaluation of the TAP device could benefit from a larger sample size and comparisons to other assay technologies of the same biomarkers. Future studies will explore monitoring of more frequent timepoints within the AD population to obtain clinical outcomes and performance data and the inclusion of more AD biomarkers for a more comprehensive panel. Additional consideration may be inclusion of biomarker-specific correction factors to account for directionally-related trending differences between TAP and VP collections. Next steps will include a clinical feasibility study to assess AD patient samples against a normal reference population.
REFERENCES
- Yourbio Health TAP Blood Collection Device and Technology: www.yourbiohealth.com
- Quanterix Simoa HD-X Technology: www.quanterix.com
- Chen GF, Xu TH, Yan Y, et al. Amyloid beta: structure, biology and structure-based therapeutic development. Acta Pharmacol Sin. 2017;38(9):1205-1235. doi:10.1038/aps.2017.28
- Leuzy A, Mattsson-Carlgren N, Palmqvist S, Janelidze S, Dage JL, Hansson O. Blood-based biomarkers for Alzheimer’s disease. EMBO Mol Med. 2022;14(1):e14408. doi:10.15252/emmm.202114408
- Lewczuk P, Łukaszewicz-Zając M, Mroczko P, Kornhuber J. Clinical significance of fluid biomarkers in Alzheimer’s Disease. Pharmacol Rep. 2020;72(3):528-542. doi:10.1007/s43440-020-00107-0
- Janelidze S, Mattsson N, Palmqvist S, et al. Plasma P-tau181 in Alzheimer’s disease: relationship to other biomarkers, differential diagnosis, neuropathology and longitudinal progression to Alzheimer’s dementia. Nat Med. 2020;26(3):379-386. doi:10.1038/s41591-020-0755-1
- Khalil M, Teunissen CE, Otto M, et al. Neurofilaments as biomarkers in neurological disorders. Nat Rev Neurol. 2018;14(10):577-589. doi:10.1038/s41582-018-0058-z
- Hol EM, Pekny M. Glial fibrillary acidic protein (GFAP) and the astrocyte intermediate filament system in diseases of the central nervous system. CurrOpin Cell Biol. 2015;32:121-130. doi:10.1016/j.ceb.2015.02.004
- Jack CR Jr et al., NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Alzheimer’s Dement. 2018 Apr;14(4):535-562.